The use of the MultiNeb® nebulizer with Agilent AVS valve systems and SPS4 autosampler with online internal calibration technique
1. Introduction
It is well known that the 6 and 7-port Advanced Valve System (AVS) reduces the cost per analysis and more than doubles the productivity of your Agilent ICP-OES. It has a positive impact on the long lifespan of consumables, instrument accessories, and plasma argon consumption by enabling switching pathways through the loading of a sample loop.
The AVS is installed between the peristaltic pump and the nebulizer on the Agilent Technologies ICP-OES instrument models 5100/5110/5800/5900.

2. Experimental
Reagents and solutions
Instrumentation
To adapt MultiNeb® nebulizer to the AVS6 valve, it is necessary to cut one of the two inlet channels to 7 cm and seal it with a 1/8 mm OD fitting as shown in Figure 1 (Part No. CN2020050, Ingeniatrics Tecnologías S.L.).

Figure 1. Connector MultiNeb AVS 6-7 (Part No. CN2020050, Ingeniatrics Tecnologías S.L).
Conventionally, the internal standard is mixed with the calibration standards and samples using a Y connection, when one inlet nebulizer in employed.
However, the novel MultiNeb® has been developed which allows a high mixing efficiency between two liquids, miscible or immiscible, since the mixing takes
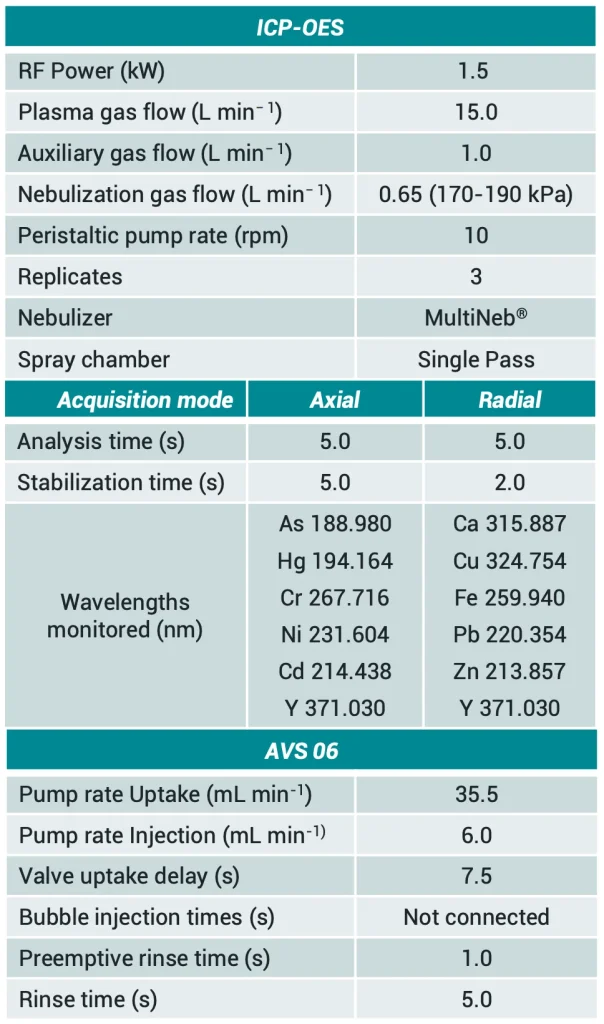
place under turbulent conditions of high pressure at the tip of the nebulizer. For internal standard introduction a tygon tubing for ISTD 0.19 mm i.d. was employed (Part No. G1820-65220, Agilent Technologies). The instrumental configuration employed, and the optimized operational conditions are shown at Table I.
Sample Preparation
A total of 20 mL of sample and 2.0 mL concentrated HNO3 and 0.5 mL of 30 % w/w H2O2 were transferred to a capped glass vials. Then, the digested samples were diluted to the final volume of 25 mL with ultrapure water. Each sample was prepared ten times.
The resulting solution was filtered through Iso-Disc poly(vinylidene difluoride) filters (25 mm diameter, 0.45 µm pore-size).
The samples were donated by a control analysis laboratory located in Huelva (Spain), collected near to an industrial area.
3. Results and discussion
Sensitivity and signal stability
The MultiNeb® nebulizer uses Flow Blurring nebulization technology instead of the traditional Venturi effect. This allows the generation of a very fine droplet aerosol with a narrow size distribution (most droplets are smaller than 10 μm), which improves efficiency over a wide range of nebulization gas flow rates, especially 0.60-0.75 L min-1 (150-250 kPa nebulization pressure).
The optimal aerosol generated by the MultiNeb® nebulizer is also more efficiently desolvated and excited in plasma, helping to improve precision values, typically less than 1% RSD, even at low sample flow rates, which also explains why it is much more sensitive than conventional nebulizers.
In order to study signal stability and plasma drift, an online internal standard correction method was utilized. The second channel of the MultiNeb® nebulizer was employed for the quantification of the analytes of interest.
However, a monitoring standard solution containing 5 µg g-1 of each element was prepared. This solution was analyzed once every 5 digested wastewater samples, in order to evaluate the stability of the signal, obtaining acceptable results for all the elements in a confidence interval of 98-102% for an analytical batch with 46 analytical determinations.
Additionally, the method detection limits (MDLs) were established by analyzing ten replicate injections of the calibration blank and multiplying the obtained standard deviation by three. The results obtained are show in Table II.

Table II. Experimental values, recovery and LOD for each monitored wavelength, as well as the RSD obtained for 10 replicates of each wastewater sample used in this application note.
Precision and reproducibility evaluation
Precision values were assessed by conducting a quality control (QC) recovery test to validate the method’s performance in real wastewater samples.
Adequate spiked levels for each element were utilized. The results of this test are presented in Table II below, which summarizes the recorded experimental values for each monitored wavelength, along with the relative standard deviation (RSD) obtained from 10 replicates of each wastewater sample used in the experimental development of this application note.
A total sequence of 30 sample determinations, 10 calibrations levels and 6 monitoring standard solution containing 5 µg g-1 of each element were analyzed in 45 minutes, approximately 1 minute of analysis time per sample.
Finally, in Figure 2 we can observe an example of the high precision, sensitivity and reproducibility previously discussed when MultiNeb® nebulizer in combination with ICP-OES instrument equipped with a AVS6 valve and an autosampler SPS4 (Agilent Technologies), for 50 µg kg-1 of Hg (194.164nm), an average of 2522.658 cps with a 0.27 % RSD.

Figure 2. Results obtained for 50 µg kg-1 of Hg (194.164 nm) using the instrumental configuration and operational conditions optimized in this Application Note.
4. Conclusions
When used together with the Agilent 5110 ICP-OES and AVS 06 valve system, the MultiNeb® nebulizer provides excellent values of sensitivity, stability and precision under the conditions described in this application note, offering an amazing testing speed and a great reproducibility for analytical batches of 46 samples with the use of an online internal standard.
The configuration presented also enables the analysis of both majority and minority elements within the same analytical batch. This approach takes approximately 1 minute per sample and maximizes the benefits of axial and radial acquisition modes offered by the Agilent ICP-OES 5110 instrument.
The conditions and configuration optimized in this study can also be extrapolated to the ICP-OES 5100, 5800 and 5900 models from Agilent Technologies.



